Diarmuid Flavin, CEO, Biosensia
Dublin-based Biosensia is developing technology to make it faster and easier to diagnose many conditions on the spot. We catch up with CEO Diarmuid Flavin to find out more about what sets the company’s technology apart in a competitive market.
Getting the right diagnosis is a key step for a patient, but many diagnostic tests can take time. Someone going to their GP often has to return or call for the results at a later stage. And in a more critical situation in hospital, any delay in diagnosis can have an impact on outcome.
‘Point-of-care’ technology seeks to slash the waiting time by analysing small volumes of patient samples on the spot and providing answers quickly.
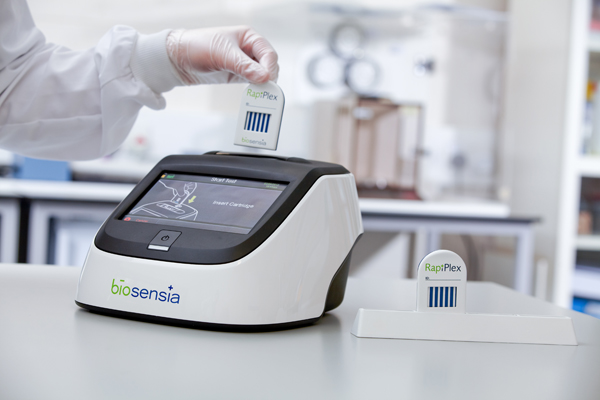
“Point of care is the ability to be able to perform what are traditionally complex immunoassays in a decentralised fashion,” explains Diarmuid Flavin. He is CEO of Biosensia, which was incorporated in 2007 to develop point-of-care technology that can handle many different sample types and tests in order to make diagnosis more patient-friendly.
Faster results
“At the moment if you present yourself at a GP’s office and you need a blood test done, what typically happens today is that the blood is taken in several tubes and sent to a central lab for testing,” says Flavin, who is based at NovaUCD. “Our technology allows you to perform the same tests right there and then. We can provide the result for the ailment that you are wishing to have diagnosed in that office during that visit time.”
The same applies to hospitals, where being able to quickly test patients coming in with suspected serious infections or heart attacks is an advantage.
“In designing the technology we wanted to be able to do what a central lab piece of kit can do with the same performance in terms of accuracy and precision of result, but within a miniaturised instrument and companion cartridge at point of care,” says Flavin.
Competitive market
Point of care is a competitive and growing market – being driven in part by the emergence of clinics in large retail chains – and there is a tremendous amount of activity in the space, he notes.
“We expect to see a measurable drift away from central labs over the next four to five years. Right now 25pc of all diagnostic testing is point of care, we expect to that to go to 30pc,” explains Flavin.
So how does Biosensia’s RapiPlex technology seek to stand out from the crowd? The platform can cope with large numbers and types of samples and tests, explains Flavin.
“Our platform technology lends itself to multiplex analysis,” he says. “The advantage of what we have done is that we have one reader and you can have multiple different products off that reader – each cartridge can provide as little as one analyte result or as many as 12 or any combination thereof.”
One of the reasons for developing the multiplex platform is to future-proof the technology, explains Flavin.
“There are a whole host of emerging biomarkers coming to the fore now, such as for kidney and liver damage and cardiovascular risk profiling. We are actively seeking and have discussions with collaboration partners around new content for the platforms.”
The Biosensia approach can also cope with many different types of samples, including whole blood serum plasma, saliva, urine, milk and stools.
“Some platforms are very good at the small volume whole blood, but they can’t accommodate large samples,” says Flavin. “We can work across a range of sample types and volumes.”
Investment
In late 2011, Biosensia announced it had secured new funding of €1.2m. The funding round was led by ACT Venture Capital, through its AIB Start-up Accelerator Fund, and included existing investors Seroba BioVentures and Atlantic Bridge.
“Our core objective in raising those funds was to push the technology forward and get approval,” explains Flavin. “Now we are about to receive our first CE mark, which we expect to have in early 2013, then we will submit our first product for US 510k approval at the same time.”
The initial approval is being sought for the RapiPlex platform using a myoglobin assay, and Flavin says Biosensia plans to develop more products, including tests for screening sexually transmitted diseases.
At the moment, around 10 people work for the company, but Flavin foresees a “significant growth phase” in the next 18 months as Biosensia ramps up to the manufacturing phase.
“The core science, reagent manufacture and the core basis of the assays themselves will be managed and manufactured from here,” he says.
Siliconrepublic.com is hosting Med Tech Focus, an initiative which over coming months will cover news, reports, interviews and videos, documenting Ireland’s leading role in one of the hottest sectors in technology.